| AAFP Feline Vaccine Guideline Explanations |
This section will try to summarize some of the AAFP vaccine guidelines. This is not a direct recommendation for your pet but intended to be used to help you understand what vaccines are available and how they are being viewed by AAFP. Please click to the right for the direct link to the AAFP guidelines.
Note: the quoted text on this page comes from the AAFP's "Summary: Vaccination in General Practice".
|
 |
| Click here for AAFP Vaccine Guidelines |
|
|
| Quick Adjuvant Information |
| With cat vaccines, there is specifically a lot of controversy over any
vaccines that contain adjuvants. An adjuvant is basically �something�
that makes vaccines work better.
Adjuvants create tissue inflammation at the site of injection and
thereby 'catch the attention' of the immune system to the vaccine.
The feline vaccine controversies are in many ways greater than the canine vaccines. There is strong evidence that adjuvants may cause cancer. AAFP states
the controversy but declines to take a stance. I try to mention where I provide additional information from AAFP, I want to fully disclose that I am not impartial on some of these vaccines, specifically I strongly recommend only non-adjuvanted vaccines. I recommend that everyone visit the AAFP site and research the vaccines on their own to gain their own insight before vaccinating their cat.
|
 |
| Click here for Vaccine Associated Sarcoma information |
|
|
| Important Vaccine Definitions |
Types of Vaccines
- Killed
- Modified Live
- Recombinant
| Vaccines are designed to 'confuse' the body's immune system into thinking that the body has an infection when actually it does not. This is how the body creates a proper immune response to help prevent infection. Currently there are four ways that vaccines work: Killed, Modified Live Virus (MLV), Recombinant Technology and Naked DNA (this last form is not used in general vaccines at this time).
Killed is the oldest method and involves taking a normal virus, chopping it up and taking out 'bits' that trigger the immune system. These trigger (antigenically stimulating) proteins are injected as the vaccine. Often killed vaccines will require the addition of something called an 'Adjuvant' which helps stimulate inflammation at the injection site to help provide an immune response. These adjuvants have been shown to have some side effects - especially in cats! See VAS article. Furthermore, it should be noted that while Killed vaccines typically will stimulate antibody formation, it does not stimulate Cell Mediated Immunity which is another important component of pet protection.
Modfied Live Virus (MLV) vaccines use an attenuated, inactive form of the virus. Since MLV acts and behaves like an actual virus, MLV often work better than killed vaccines as it stimulates Cell Mediated Immunity. However, since it is a truly live virus of a disease that we are trying to prevent, there is a small risk that the inactive virus can revert into the true form of the virus. For this reason, there is NO MLV Rabies vaccine made. As a general rule, 'reversion' to a virulent viral form is very, very rare.
Recombinant Technology is relatively new and been shown to have some wonderful advantages. It uses a live virus from another species, for example a canary pox virus, so that true infection is not possible. It then takes only the portions of the RNA of the virus that we wish to protect against and places them within the canary virus (the canary pox virus is a DNA virus. Thus the RNA portions of the virus (canine distemper, rabies, feline leukemia) is reverse transcribed into DNA so that it can be placed within the virus. Just a technical point.) The only portions of the original virus RNA that is used are those that make the proteins that the body's will recognize. As the rest of the virus RNA is never used, there is not ability for a complete virus to form. The live canary virus will then be used in the vaccine, and cause replication of these proteins within the local macrophages and other cells. The proteins become expressed on the cell surface leading to both a strong humoral and cell mediated immunity.
Benefits of Recombinant Technology.
One of the great benefits of Recombinant Technology is that the vaccine cannot become inactivated by maternal antibodies like the other vaccines. This is very important for puppies and kittens who are under 16 weeks of age. Under this age, the antibodies that the pets have gotten from their mother can inactivate the Killed and MLV vaccines and render the vaccination useless. Thus Recombinant Technology vaccines may be more appropriate for younger animals.
With the concern over VAS (Vaccine Associated Sarcomas), many clinicians are favoring recombinant vaccines. Sarcomas can be deadly, and may occur up to 8 years after the vaccine was given. This potential risk of sarcoma is one of the main reasons why Merial has developed the feline Recombinant series of vaccines.
|
| AAFP Feline Core Vaccines |
Combo Vaccine
| The basic combo vaccine for cats includes: 1) Panleukopenia Virus (FPV) which is very similar to the canine parvo virus, 2) Feline Herpes-1 (FHV-1) which causes feline upper respiratory infections and 3) Feline Calicivirus (FVC) which also causes feline upper respiratory infections. Panleukopenia is also known as Feline Distemper but it bears no resemblance to the canine distemper virus.
Note: Feline Chlamydia is sometimes included in the 'Combo' vaccine to make a 4 in 1. It is explained further down.
Initial Kitten Vaccination: "Begin as early as 6 weeks of age, then every 3-4 weeks until 16 weeks of age." If the unvaccinated kitten is older than 16 weeks of age, then AAFP recommends "2 doses, 3-4 weeks apart"
Revaccination: "A single dose is given 1 year following the last dose of the initial series, then no more frequently than every 3 years."
3 in 1 feline combo vaccine forms:
- Injectable
- Modified Live Virus (MLV), non-adjuvanted
- Killed, Adjuvanted (please see VAS article)
- Killed, non-adjunaved
- Intranasal
- Modified Live Virus (MLV), non-adjuvanted
Important Notes
- Young kittens
- The Modified Live Virus (MLV) Panleukopenia vaccine should NOT be used in kittens under 4 weeks of age. There has been associated cerebellar hypoplasia (a debilitating and irreversible brain disease).
- Lack of a Recombinant form
- There is no recombinant form of this. As MLV form is effective and non-adjuvanted, this is the recommended vaccine.
- Intranasal Vaccine Concerns (not from AAFP guidelines)
- There is some controversy here. The duration of
immunity is not clear. It might not be safe to vaccinate with less
frequency than every year. Furthermore there is speculation that the
panleukopenia portion may not be as effective as the injectable form.
The normal route of infection for panleukopenia is also not nasal.
Until there is more information, it is recommended to use the
injectable non-adjuvanted form of this vaccine.
|
Rabies Vaccine
| Initial Kitten Vaccination: "Administer a single dose as early as 8 or 12 weeks of age depending on the product label." There may also be state regulations that need to be followed for the minimum age of vaccination.
Revaccination: For the 1 year labeled vaccines, annual vaccination is required. The 3-year vaccine may be used every three years or "as required by State or local ordinance for 3 year."
Feline Rabies Forms:
- Recombinant, Non-Adjuvanted. Labeled for one year only. The author's recommendation.
- 1-year killed, adjuvanted
- 3-year killed, adjuvanted
Important Notes:
- "Any rabies vaccine can be used for revaccination, even if the product is not the same brand or type of product previously administered." For this reason, if your cat has previously been receiving adjuvanted vaccines, you may want to consider switching him or her over to the Recombinant vaccine.
|
| AAFP Feline Non-Core Vaccines |
Feline Leukemia Vaccine
| This vaccine should almost be in both sections of Core and Non-Core. For kittens, this should be, in my humble opinion, considered 'Core' or a 'Must Have'. AAFP states, "FeLV vaccination is highly recommended for all kittens."
Adult cats, vaccinated or not, appear to have some natural immunity to the leukemia virus. There is controversy as to which cats should receive it and who should not. Many clinicians do not recommend vaccinating strictly indoor cats who do not come in contact with outdoor cats.
"Since adult cats are highly naturally resistant to FeLV and because
vaccination should provide them with some additional protection, I
would continue to vaccinate the in/out cats." - Alice M. Wolf, DVM, DACVIM, DABVP
Initial Kitten Vaccination: "Administer an initial dose as early as 8 - 12 weeks of age, depending on product; a second dose should be administered 3-4 weeks later."
Revaccination: Yearly to those cats determined to be at risk of exposure.
FeLV Vaccine Forms:
- Recombinant, non-adjuvanted
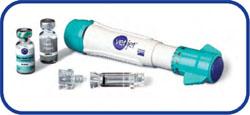
- This vaccine is administered with a Transdermal VetJet Device. It is given as a needless injection.
- In Europe, there is a recombinant subcutaneous injectable form. There is none currently available in the United States.
- Killed, adjuvanted
- This vaccine may have a high rate of VAS formation
|
Link to the Merial Recombinant FeLV Vaccine. Click to the right.
Note: If your cat has been given a Feline Leukemia Vaccine without the device pictured to the right, then it was not a Recombinant Non-Adjuvanted form.
|
 |
| Click here for the Recombinant FeLV |
|
|
Chlamydia Vaccine
| Chlamydophia felis was previously called Chlamydia.
Initial Kitten Vaccination: As it is only available within Combination Vaccines - See Combo vaccine.
Revaccination: "Annual booster is indicated for cats with sustained exposure risk." See notes below.
Chlamydophila forms:
- Avirulent (non-disease forming) live, non adjuvanted
- Killed Adjuvanted
AAFP Comment
- "Vaccination reserved as part of a control regime for cats in multiple-cats environments where infections associated with clinical disease have been confirmed."
Important Notes (Not sourced from AAFP):
- Chlamydiophila vaccine may have a higher rate of vaccine reaction than many other vaccines.
- "Chlamydophylia isn't a problem for household pets. This vaccine might
be useful on a short term basis in catteries or shelters having a
respiratory disease outbreak due to confirmed infection with this
agent. Like all bacterins, if you use Chlamydophia, it should be
repeated yearly if you want reasonable protection. Since we don't want
or need to give the FVRCP yearly and there is no "stand alone"
Chlamydophila product to my knowledge, that makes its routine use
somewhat problematic." - Alice M. Wolf, DVM, DACVIM, DABVP
|
Bordetella Vaccine
| Bordetella bronchiseptica is the bacteria that causes Kennel Cough. While it is common in dogs, its prevalence in cats is considered low.
Initial Kitten Vaccination: "Administer a single dose inranasally as early as 8 weeks of age"
Revaccination: "Annual booster is indicated for cats with sustained risk"
Bordetella forms:
- Intranasal avirulent (non-disease forming) live, non-adjuvanted
AAFP Comments
- Some specific situations for its use include, "prior to confinement in multiple-cat environments such as rescue shelters, boarding facilities or cattteries where bordetellosis has been confirmed."
- Do not give as an injection. It "may cause a severe adverse reaction that can cause the death of the cat."
|
FIV Vaccine
| Feline Immunodeficiency Virus (FIV) is a virus that suppresses the immune system. It is similar to HIV (Human Immunodeficiency Virus) but it is NOT TRANSMISSIBLE TO HUMANS. It is a not a zoonotic disease.
This vaccine is contoversial. Many clinicians, including myself, have severe reservations on this vaccine.
Initial Kitten Vaccination: "The initial dose is administered as early as 8 weeks of age; 2 subsequent doses should be administered at an interval of 2-3 weeks"
Revaccination: Annually for those cats at risk.
Form
AAFP Comments
- "FIV vaccine should be restricted to cats at high risk of infection. For example, outdoor fighting cats and FIV-unifected cats living with FIV-infected cats."
Important Concerns (not all sourced from AAFP)
- First of all it is adjuvanted.
- Second, it can interfere with testing; meaning that vaccinated cats will test positive for FIV. It can be difficult to establish whether the pet has FIV or was previously vaccinated, This can pose a serious concern to cats that are adopted into new homes. At one year after vaccination 30% will be seronegative (not cause a false positive). Data for the other 70% and when they become seronegative is not completely understood. Thus a cat who is adopted for several years, not vaccinated for FIV by the new owners, and tests positive for FIV may very well still not have FIV but just have antibodies secondary to prior vaccination. For owners with multiple cat households, this can be frustrating and scary
- Thirdly, there are five clades (like strains) of FIV. The virus was designed for clades A and D but only fully tested for clade A. The vaccine was not tested for clade B which is a major subtype in the United States. There is some evidence that the vaccine might not protect against this clade. Also one study showed that the efficacy against clade A (the one it was both designed and tested for) is only 82%.
|
| AAFP - "Non Generally Recommended" |
FIP Vaccine
| Feline Infectious Peritonitis (FIP) is a deadly form of the feline coronavirus. The coronavirus infection is common and may be present in as much as 70% of high cat density locations. Most of the cats with feline coronavirus will NOT develop FIP.
Why it is not recommened: It is thought only to help cats that do not have the feline corona virus. As many cats do, the vaccination will not help corona infected cats. Also there is evidence that the cats that have feline corona virus conversion to FIP will have worse disease if vaccinated with this vaccine. "I don't recommend this vaccine at all. It has not been shown to be
efficacious and in studies by Dr. Scott at Cornell, may sensitize cats
to worse disease." - Alice M. Wolf, DVM, DACVIM, DABVP. 'Cats share coronaviruses but the reason they develop FIP is an
individual event (pathogenic mutation) in each individual cat. No
vaccine will prevent cats from carrying coronavirus." - Alice M. Wolf, DVM, DACVIM, DABVP.
|
Giardia Vaccine
| Giardia is a protozoal parasite that affects many mammals including
dogs, cats and humans. While the strain (for Giardia, called Molecular
Assemblages) that affect humans is usually not the molecular assemblage
seen in dogs, canine Giardiasis can be transfered to humans.
Why it is Not Recommended: "There is insufficient studies available to support the role of Giardia vaccination in preventing clinical disease in cats." While The dog Giardia vaccine is also controversial, there is some information to support canine use. There is less evidence supporting its use in cats.
|
|